Dietrich lab
The Laboratory of Tumor Immunology, headed by Prof. Pierre-Yves Dietrich has been active for many years in glioma immunology. In the last decade, our group contributed to the understanding of important new concepts in the field of brain tumor immunology and in the development of immunotherapy for patients with brain cancer. The current research group is comprised of junior and senior research scientists developing individual projects, assisted by skilled research technicians. ...
Research projects
Brain Tumor Immunology
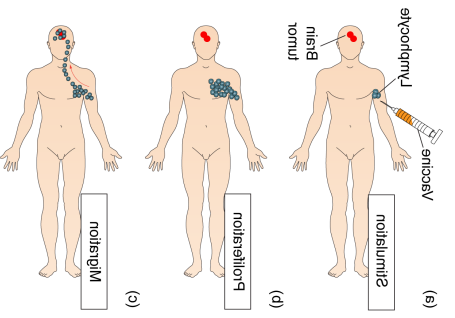
(i) T cell trafficking to the brain (Calzascia 2003, 2008)
(ii) Identification of glioma antigens (Dutoit Brain 2012)
(iii) Therapeutic vaccines (Hilf Nature 2019, Migliorini Neuro oncol 2019)
A key step in the development of immunotherapy for glioma was the identification of a set of glioma antigens by the Dietrich group in 2012, and the initiation of several clinical trials based on these findings. These studies showed that CD4 and CD8 T cell responses to the vaccines can be elicited. Further approaches of combining these vaccines with other treatment modalities are currently being tested.
This identification of brain tumor antigens in our laboratory was directly translated into the clinic through a clinical trial of peptide vaccination using our targets, which started in Geneva in 2013. This vaccine, given to patients already suffering from the disease, specifically stimulates tumor-reactive T lymphocytes present in the patient’s blood that patrol the body to find and kill tumor cells. After having vaccinated 19 patients, we are now analyzing the immune responses elicited by the vaccine and are following the patients’ clinical evolution. This has provided us with important information to understand the effect of the vaccine on the patients’ immune system and disease. Importantly, a follow-up trial is currently ongoing at HUG, which combines the present peptide vaccine with (i) an anti PD1 antibody (pembrolizumab) that allows sustained activation of T cells, thereby improving vaccine efficacy, and (ii) a modulator of the microenvironment (TLR agonist). This work has led to the concept of personalized peptide vaccines and has allowed us to design better vaccines for patients suffering from glioma.
Colorectal Tumor Immunology
Rectal cancer is one of the ten most common cancers worldwide. Current curative treatment sequences use radiotherapy or chemoradiotherapy followed by surgery. Although curative, this combined approach is associated with important morbidity due to the loss of continence and an important risk of relapse. A major obstacle in the development of effective therapies is posed by the intrinsic and extrinsic biological heterogeneity of these tumors.
Our group focuses on studying the tumor immune microenvironment in rectal cancer and its modifications following radiotherapy or chemoradiotherapy with the aim to uncovering ways to harness it for therapy.
Currently, we are working on four different projects:
- Capturing cellular dynamics in rectal cancer using scRNA sequencing:
Our study aims at bridging this knowledge gap by generating a single-cell atlas of RC tumor cells and their surrounding immune and stromal cells, as well as exploring cell-specific transcriptional changes related to specific therapeutic interventions.
- Tackling immune exclusion in microsatellite stable rectal cancer:
By applying a combination of in vitro and in vivo rectal cancer models, ‘omics’ approaches and advanced imaging techniques, we aim at developing alternative approaches to promote de novo T cell activation and infiltration into the tumor microenvironment of microsatellite stable rectal cancer and other non-inflamed tumors.
- Breaking barriers to successfully treat rectal cancer - a clinical trial:
Our clinical trial (PEMREC) is the first study evaluating a novel therapeutic approach that combines radiotherapy with an anti-PD1 antibody. This study could lead to a major breakthrough in the treatment of rectal cancer.
- Explore functional states and spatial interactions between cell types using multiplexed-IF and advanced image analysis:
Here, we aim at developing a multiplexed immunofluorescent technology to FFPE tissue along with a software for digital pathology to characterize the tumor microenvironment of rectal cancer.

Latest publications
Study protocol of a phase II study to evaluate safety and efficacy of neo-adjuvant pembrolizumab and radiotherapy in localized rectal cancer.
Corrò C, Buchs NC, Tihy M, (...), Dietrich PY, Ris F, Koessler T
BMC cancer – 2022 Jul 15
Linking unfolded protein response to ovarian cancer cell fusion.
Yart L, Bastida-Ruiz D, Allard M, (...), Dietrich PY, Petignat P, Cohen M
BMC cancer – 2022 Jun 7
Une période de terreur et de défis : quand verrons-nous ce monde meilleur ?
Dietrich PY, Aapro M, Peters S
Revue medicale suisse – 2022 May 18
Team

Pierre-Yves Dietrich
MD, Head Department of Oncology, Geneva University Hospital, Co-Director SCCL, HUG/UNIGE
DietrichLabSelected Publications
Patients’ experiences with cancer care in Switzerland: Results of a multicentre cross-sectional survey.
Arditi C, Eicher M, Colomer-Lahiguera S, (...), Duchosal M, Peters S, Peytremann-Bridevaux I
European journal of cancer care – 2022 Sep 21
Allogeneic CAR T Cells: An Alternative to Overcome Challenges of CAR T Cell Therapy in Glioblastoma.
Martínez Bedoya D, Dutoit V, Migliorini D
Frontiers in immunology – 2021 Mar 3
Single-Cell Analyses Identify Brain Mural Cells Expressing CD19 as Potential Off-Tumor Targets for CAR-T Immunotherapies.
Parker KR, Migliorini D, Perkey E, (...), Chang HY, Posey AD Jr, Satpathy AT
Cell – 2020 Sep 21
Phase I/II trial testing safety and immunogenicity of the multipeptide IMA950/poly-ICLC vaccine in newly diagnosed adult malignant astrocytoma patients.
Migliorini D, Dutoit V, Allard M, (...), Walker PR, Patrikidou A, Dietrich PY
Neuro-oncology – 2019 Jul 11
Actively personalized vaccination trial for newly diagnosed glioblastoma.
Hilf N, Kuttruff-Coqui S, Frenzel K, (...), Sahin U, Dietrich PY, Wick W
Nature – 2018 Dec 19
Glycan-directed CAR-T cells.
Steentoft C, Migliorini D, King TR, (...), Mandel U, June CH, Posey AD Jr
Glycobiology – 2018 Sep 1
Glut3 Addiction Is a Druggable Vulnerability for a Molecularly Defined Subpopulation of Glioblastoma.
Cosset É, Ilmjärv S, Dutoit V, (...), Weis SM, Mischel PS, Cheresh DA
Cancer cell – 2017 Nov 30
CAR T-Cell Therapies in Glioblastoma: A First Look.
Migliorini D, Dietrich PY, Stupp R, (...), Linette GP, Posey AD Jr, June CH
Clinical cancer research : an official journal of the American Association for Cancer Research – 2017 Nov 20
Hypoxia and antitumor CD8+ T cells: An incompatible alliance?
Vuillefroy de Silly R, Dietrich PY, Walker PR
Oncoimmunology – 2016 Sep 9
Exploiting the glioblastoma peptidome to discover novel tumour-associated antigens for immunotherapy.
Dutoit V, Herold-Mende C, Hilf N, (...), Weinschenk T, Singh-Jasuja H, Dietrich PY
Brain : a journal of neurology – 2012 Mar 14
Prevention of CNS relapse in diffuse large B-cell lymphoma.
Kridel R, Dietrich PY
The Lancet. Oncology – 2011 Sep 18
Promising survival for patients with newly diagnosed glioblastoma multiforme treated with concomitant radiation plus temozolomide followed by adjuvant temozolomide.
Stupp R, Dietrich PY, Ostermann Kraljevic S, (...), de Tribolet N, Mirimanoff RO, Leyvraz S
Journal of clinical oncology : official journal of the American Society of Clinical Oncology – 2002 Mar 1
Tumor expression of Fas ligand (CD95L) and the consequences.
Walker PR, Saas P, Dietrich PY
Current opinion in immunology – 1998 Oct
Related news
Events

Leçon d’adieu Prof. Pierre-Yves Dietrich | November 24th
Events

