Merten lab
The laboratory of biomedical microfluidics (LBMM) develops novel approaches for drug discovery and personalized cancer therapy. Having a strong translational focus, we collaborate with pharma industry, foster entrepreneurship and pave the way for first clinical trials. In line with this, we run a tech development lab on the EPFL campus, a translational lab in the AGORA cancer research center (with direct access to patient material) and we founded a startup company making the results of our research widely available. The lab is very interdisciplinary and includes people with various backgrounds ranging from biology, genomics and bioinformatics to engineering and physics. ...
Research projects
As a common theme we use microscopic droplets as miniaturized reaction vessels. This offers some unique advantages:
Drastically increased throughput (processing up to a million samples an hour).
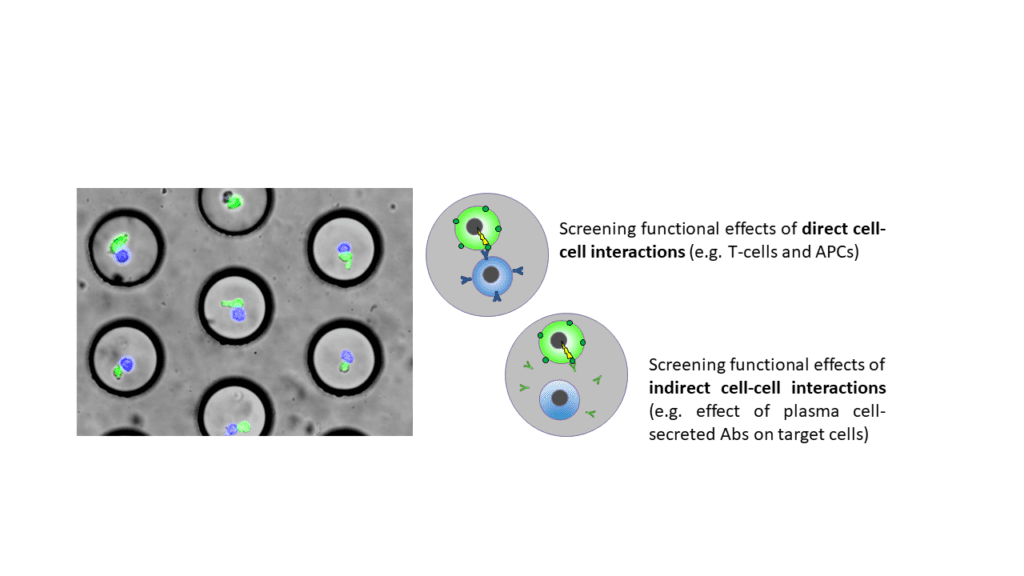
Possibility of performing cell-cell interaction screens at the single-cell level (phenotypic and genotypic assays).
Low sample consumption, enabling the use of patient material.
Having a comprehensive droplet microfluidic toolbox at hand (and expanding it continuously), we are now focusing on the following research themes:
Therapeutic antibodies and T-cells
Droplet based microfluidics enables to screen a large fraction of the murine and human immune repertoire in a single experiment (e.g. El Debs et al., PNAS 2012, Chaipan et al., Cell Chemical Biology 2017, Shembekar et al., Cell Reports 2018). We are actively exploiting this for novel therapeutic approaches and have founded a biotech startup company translating our results (www.veraxa.de).
Personalised medicine
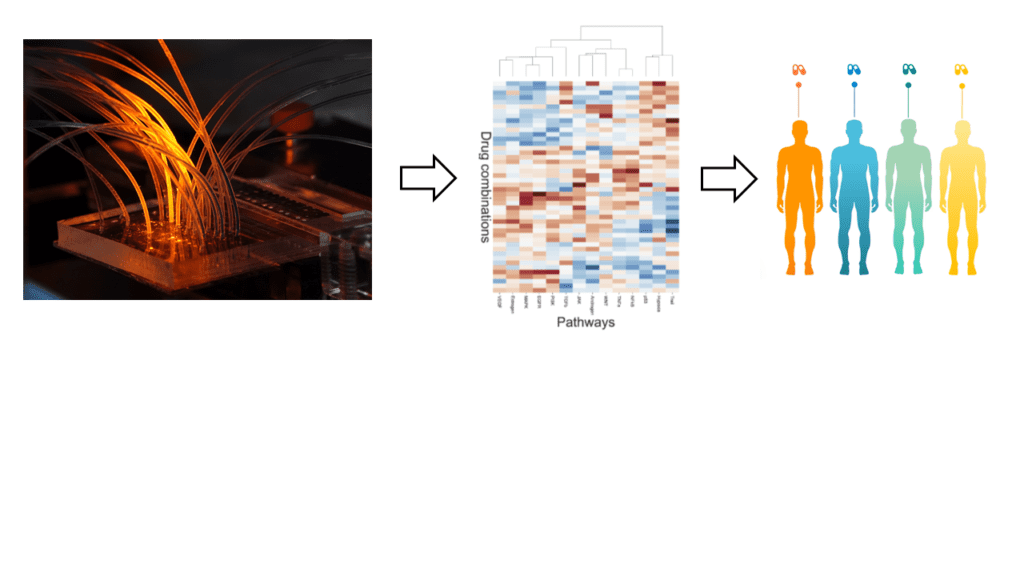
We use microfluidic devices to test drug combinations directly on solid human tumor samples and to predict optimal therapies (e.g. Eduati et al., Nature Communications 2018, Mathur et al, bioRxivs 2021). Current efforts focus on the development of highly multiplexed genetic and imaging-based readouts. In parallel, we are constructing next generation instruments for first clinical trials and prepare the launch of another startup, making our personalized cancer therapy approaches available to the public (TheraMe! Consortium www.besttherapyforme.com).
Genomics
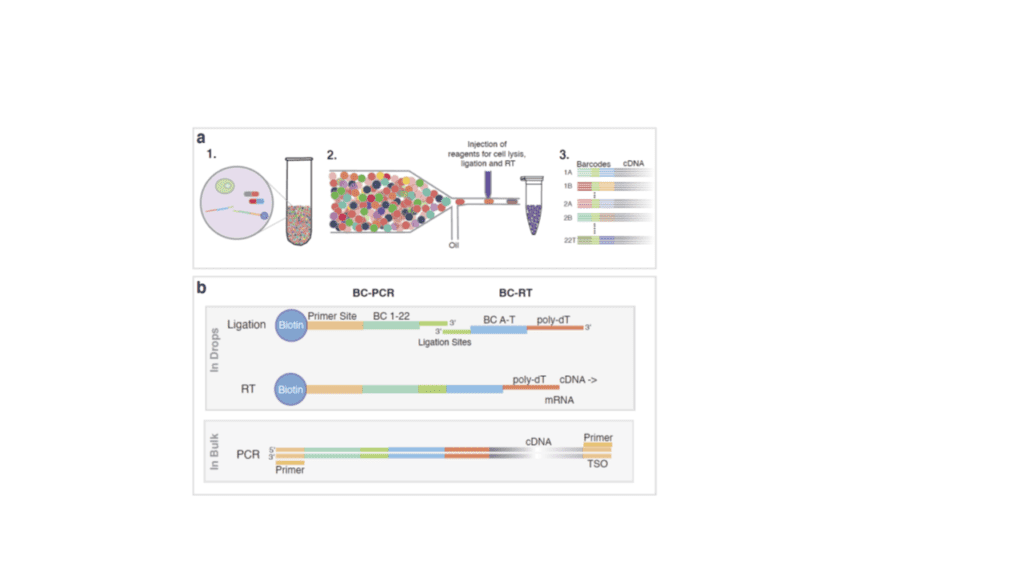
We are actively developing microfluidic modules for single-cell barcoding and sequencing. This will enable the study of the mechanisms of drug resistance in great detail. In the past we have also developed microfluidic modules for the shearing of genomic DNA into fragments that are short enough for next generation sequencing. This technology has been licensed and is now commercially available world-wide, e.g. in form of the bioruptor.
Microfluidic Technology Development
We are continuously developing microfluidic chips with improved functionality (e.g. fully automated droplet sorting and fusion, multiplexed-multicolor-multichannel sorting), design new hardware (e.g. for impedance measurements, temperature control, etc.) and program new control software (mostly LabVIEW & Python). The overarching goal is to make the technology more robust, more automated (= accessible to non-specialists), faster and more versatile.

Latest publications
Recovery of phenotypically sorted cells using droplet-digital microfluidics.
Deng Z, Perry JM, Weiss M, (...), Autour A, Merten CA, Shih SCC
Lab on a chip – 2025 Jul 22
Targeted barcoding of variable antibody domains and individual transcriptomes of the human B-cell repertoire using Link-Seq.
Hu H, Zhou F, Ma X, (...), Benes V, Cox RJ, Merten CA
PNAS nexus – 2025 Jan 9
Fluorescence-activated droplet sequencing (FAD-seq) directly provides sequences of screening hits in antibody discovery.
Autour A, Merten CA
PNAS – 2024 Sep 6
Screening for drivers of SARS-CoV-2 uptake.
Autour A, Merten CA
Nature biomedical engineering – 2023 Dec 29
Large scale microfluidic CRISPR screening for increased amylase secretion in yeast.
Johansson SA, Dulermo T, Jann C, (...), Roy KR, Wei W, Steinmetz LM
Lab on a chip – 2023 Jul 24
Team

Other members
Selected Publications
Enrichment of gut microbiome strains for cultivation-free genome sequencing using droplet microfluidics.
Pryszlak A, Wenzel T, Seitz KW, (...), Benes V, Bork P, Merten CA
Cell reports methods – 2022 Jan 24
Combi-Seq: Multiplexed transcriptome-based profiling of drug combinations using deterministic barcoding in single-cell droplets.
L Mathur, B Szalai, R Utharala, M Ballinger, JJM Landry, M Ryckelynck, View ORCID ProfileV Benes, J Saez-Rodriguez, CA Merten
bioRxiv – 2021 September 17
Patient-specific logic models of signaling pathways from screenings on cancer biopsies to prioritize personalized combination therapies.
Eduati F, Jaaks P, Wappler J, (...), Merten CA, Garnett MJ, Saez-Rodriguez J
Molecular systems biology – 2020 Jun
Targeted Perturb-seq enables genome-scale genetic screens in single cells.
Schraivogel D, Gschwind AR, Milbank JH, (...), Merten CA, Velten L, Steinmetz LM
Nature methods – 2020 Jun 1
Microfluidics as an Enabling Technology for Personalized Cancer Therapy.
Mathur L, Ballinger M, Utharala R, Merten CA
Small (Weinheim an der Bergstrasse, Germany) – 2019 Nov 20
Antibiotics-induced monodominance of a novel gut bacterial order.
Hildebrand F, Moitinho-Silva L, Blasche S, (...), Patil KR, Benes V, Bork P
Gut – 2019 Jan 18
A microfluidics platform for combinatorial drug screening on cancer biopsies.
Eduati F, Utharala R, Madhavan D, (...), Cramer T, Saez-Rodriguez J, Merten CA
Nature communications – 2018 Jun 22
Modulation of Phase Shift between Wnt and Notch Signaling Oscillations Controls Mesoderm Segmentation.
Sonnen KF, Lauschke VM, Uraji J, (...), François P, Merten CA, Aulehla A
Cell – 2018 Feb 22
Single-Cell Droplet Microfluidic Screening for Antibodies Specifically Binding to Target Cells.
Shembekar N, Hu H, Eustace D, Merten CA
Cell reports – 2018 Feb 20
Microfluidic single-cell technology in immunology and antibody screening.
Seah YFS, Hu H, Merten CA
Molecular aspects of medicine – 2017 Sep 23
Single-Virus Droplet Microfluidics for High-Throughput Screening of Neutralizing Epitopes on HIV Particles.
Chaipan C, Pryszlak A, Dean H, (...), Benes V, Griffiths AD, Merten CA
Cell chemical biology – 2017 May 25
Microfluidic train station: highly robust and multiplexable sorting of droplets on electric rails.
Frenzel D, Merten CA
Lab on a chip – 2017 Mar 14
Efficient cell pairing in droplets using dual-color sorting.
Hu H, Eustace D, Merten CA
Lab on a chip – 2015 Aug 27
Dynein-Based Accumulation of Membranes Regulates Nuclear Expansion in Xenopus laevis Egg Extracts.
Hara Y, Merten CA
Developmental cell – 2015 May 21
Functional single-cell hybridoma screening using droplet-based microfluidics.
El Debs B, Utharala R, Balyasnikova IV, Griffiths AD, Merten CA
Proceedings of the National Academy of Sciences of the United States of America – 2012 Jul 2
Related news
Events
🌱 Third AGORA Sustainability Day | May 21st
Events
🌱 Second AGORA Sustainability Day | May 21st
Events

AGORA PRS Seminar | December 17th
Events

AGORA PRS | December 19th
Events

AGORA PRS | December 20th
Events







