The Patient Lab
The Patient Lab aims to ensure the inclusion of patient and public partners in cancer research for the development of healthcare systems that respond to today’s and tomorrow’s needs and challenges. Our objective is to support researchers and patient/public partners all along their research projects and to provide advice on best practices to ensure genuine partnership. ...
Research projects
Our mission & objectives
As affiliate of the Swiss Cancer Center Leman (SCCL), the Patient Lab innovates and promotes a transdisciplinary partnership with cancer patients, caregivers, and public representatives at all stages of research and development of new or improved models of support and care.
Affiliating the Patient Lab with the existing SCCL research & clinical platforms aims to
- empower each other's innovation through cutting edge interactions between researchers, patients and the public
- improve public and patient involvement in research activities, and thus their adherence to studies
- develop and enhance joint PPI capacities
- enable the development of transdisciplinary oncology research projects that deliberately and systematically involve public and patients, and lead to quality improvement initiatives in clinical care
Initially, the Patient Lab focuses on the development of Patient-Reported Outcomes Measures (PROMs) and Patient-Reported Experience Measures (PREMs) in cancer research and care.


The SCCL-PPI model
The SCCL-PPI model has been developed based on a literature review that was conducted to identify existing frameworks in cancer research internationally.
Nine models from seven different countries were taken into consideration - leading us to propose a model that is adapted to the specific needs of our local context: French-speaking Switzerland.
To learn more about the development of the SCCL-PPI model, we invite you to take a look at our website as well as the article we published in the Revue Médicale Suisse.
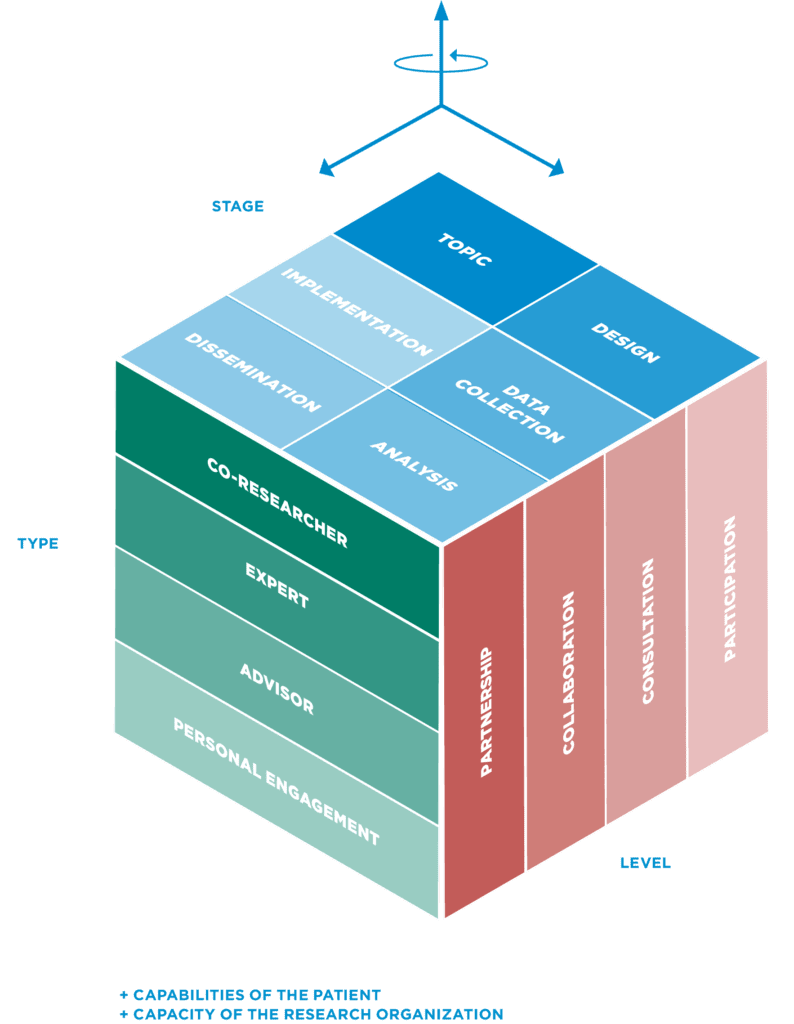
STAGES: Patients and the public can be involved at all stages of the research process, and in one or more phases of a research project .
LEVELS: In the context of the SCCL-PPI model, and based on the type of research envisioned, we define 4 different levels of patient involvement in research.
TYPES & ROLES: Patients' roles vary according to the contributions a patient is able to offer. To use patient knowledge to the best effect, ways must be found to convert the diversity of individual experience into the closest representation of a typified patient perspective. The patient voice must be a blend of collective and personal experiences. A way to overcome this is to include more than one patient and covering more than one type of involvement.
CAPABILITIES & CAPACITY: Furthermore, the decision regarding the stage, level, and type of involvement requires a careful evaluation of the capabilities of the patients as well as the capacity of the research group or organization.

Latest publications
Validation of the newly developed Advanced Practice Nurse Task Questionnaire: A national survey.
Beckmann S, Schmid-Mohler G, Müller M, (...), Eicher M, Ullmann-Bremi A, Petry H
Journal of advanced nursing – 2023 Jun 27
Patient-reported experiences of cancer care related to the COVID-19 pandemic in Switzerland.
Colomer-Lahiguera S, Canella C, Giacomini S, (...), Witt CM, Eicher M, Ribi K
Supportive care in cancer : official journal of the Multinational Association of Supportive Care in Cancer – 2023 Jun 22
A comparative study of social cognition in epilepsy, brain injury, and Parkinson’s disease.
Bauer J, Steiger BK, Kegel LC, (...), König K, Baumann-Vogel H, Jokeit H
PsyCh journal – 2023 May 1
Big Data in Oncology Nursing Research: State of the Science.
Harris CS, Pozzar RA, Conley Y, (...), Kober KM, Miaskowski C, Colomer-Lahiguera S
Seminars in oncology nursing – 2023 Apr 19
Team

Manuela Eicher
Associate Professor, Academic Co-Lead of the Patient lab, Director of the Institute of Higher Education and Research in Healthcare (IUFRS), University of Lausanne (UNIL), Nurse Research Consultant, Department of Oncology (DO), Lausanne University Hospital (CHUV)
@EicherManuelaOther members

Alfredo Addeo
Full Professor, Academic Co-Lead of the Patient lab, Head of the Oncology Service, Department of Oncology (DO), HUG
Selected Publications
Development of an eHealth-enhanced model of care for the monitoring and management of immune-related adverse events in patients treated with immune checkpoint inhibitors.
da Silva Lopes AM, Colomer-Lahiguera S, Darnac C, (...), Addedo A, Michielin O, Eicher M
Supportive care in cancer : official journal of the Multinational Association of Supportive Care in Cancer – 2023 Jul 22
Patient and public involvement in cancer research: A scoping review.
Colomer-Lahiguera S, Steimer M, Ellis U, (...), Tompson M, Corbière T, Haase KR
Cancer medicine – 2023 Jun 16
Patient-reported outcomes in adoptive cell-therapy trials: mind the gap.
Ghisoni E, Morotti M, Colomer-Lahiguera S, (...), Coukos G, Trueb L, Di Maio M
Journal for immunotherapy of cancer – 2022 Dec 18
Patients’ experiences with cancer care in Switzerland: Results of a multicentre cross-sectional survey.
Arditi C, Eicher M, Colomer-Lahiguera S, (...), Duchosal M, Peters S, Peytremann-Bridevaux I
European journal of cancer care – 2022 Sep 21
A Narrative Review on the Collection and Use of Electronic Patient-Reported Outcomes in Cancer Survivorship Care with Emphasis on Symptom Monitoring.
van den Hurk CJG, Mols F, Eicher M, (...), Howell D, Basch EM, van de Poll-Franse LV
Current oncology (Toronto, Ont.) – 2022 Jun 17
[Patient involvement in oncology research – Development of a reference framework in French-speaking Switzerland].
Colomer-Lahiguera S, Corbière T, Reeb-Landry F, (...), Ninane F, Addeo A, Eicher M
Revue medicale suisse – 2022 Mar 30
Patient-reported outcomes for monitoring symptomatic toxicities in cancer patients treated with immune-checkpoint inhibitors: A Delphi study.
Da Silva Lopes AM, Colomer-Lahiguera S, Mederos Alfonso N, (...), Addeo A, Michielin O, Eicher M
European journal of cancer (Oxford, England : 1990) – 2021 Sep 15
Patient-reported outcome instruments used in immune-checkpoint inhibitor clinical trials in oncology: a systematic review.
Colomer-Lahiguera S, Bryant-Lukosius D, Rietkoetter S, (...), Orcurto A, Juergens R, Eicher M
Journal of patient-reported outcomes – 2020 Jul 16
Adapting a peer-led self-management program for breast cancer survivors in Switzerland using a co-creative approach.
Schmidt F, Ribi K, Haslbeck J, (...), Urech C, Holm K, Eicher M
Patient education and counseling – 2020 Apr 17
Related news
Events
AGORA Sustainability Day
Events

Les coulisses de la recherche | June 6th
Events

Les coulisses de la recherche | March 7th
Events

Les coulisses de la recherche | November 16th
Events







